The Role of Supplementary Material in Journal Articles: Surveys of Authors, Reviewers, and Readers
Abstract
Amy Price,1,2 Sara Schroter,1 Mike Clarke,3,4,5 Helen McAneney5
Objective
Many journals allow or require authors to submit supplementary material for consideration when their manuscript is going through the editorial process and for possible publication with the article. We explore the value and role of supplementary material in journal articles from the perspective of authors, peer reviewers, and readers.
Design
Among authors and peer reviewers of research submissions to 17 BMJ Publishing Group journals, we randomly allocated two-thirds of each group to receive an author and reviewer survey, respectively, and the remaining third of each group to receive a reader survey. In November 2016, participants completed an online survey from the perspective of their allocated role to provide information about their use of specific types of supplementary material (study protocol, data collection or extraction forms, data tables and figures, completed reporting guideline checklists and flow diagrams, interview transcripts, raw study data). Survey questions asked about who each portion of the material is most useful to; the expected use of materials by authors, reviewers and readers; the preferred option for accessing supplementary material; and if and where supplementary material should be published.
Results
Among 20,340 surveyed authors and peer reviewers, we received 2872 (14%) responses (819 [12%] from authors, 1142 [17%] from peer reviewers, and 911 [14%] from authors and reviewers responding as readers). Most authors (711 of 819 [87%]) reported submitting at least 1 type of supplementary material with their most recent manuscript, 95% (1086 of 1142) of reviewers reported seeing supplementary material in an article they reviewed at least sometimes (ie, not never or almost never), and 79% (724 of 911) of readers reported that the article they read most recently included supplementary material. Additional data tables were the most common supplementary material type submitted or seen (authors, 74%; reviewers, 89%; readers, 67%). A majority in each role indicated additional data tables were most useful to readers (58%-72%), while fewer indicated they were most useful to peer reviewers (19%-34%) and journal editors (3%-4%) (Table). Patterns of opinion were opposite for reporting guideline checklists. All 3 groups favored publication of additional data tables and figures on the journal’s website (80%-83%), with less than 4% of each group reporting that availability was not needed. Less than one-fourth of respondents in each group said that raw study data should be available on the journal’s website (16%-23%), and 24% to 33% said that these materials should not be made available.
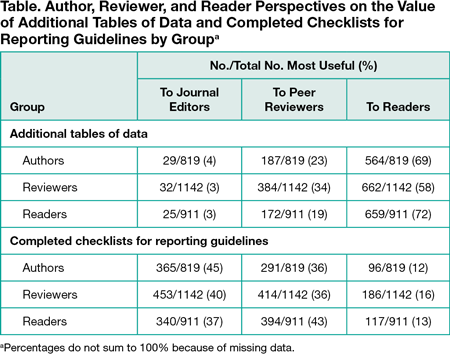
Conclusions
Authors, peer reviewers, and readers favor access to supplementary tables and figures over completed reporting checklists or raw data. These findings may help journals to consider the roles, resource costs, and strategic placement of supplementary materials for optimal usage.
1The BMJ, London, UK, dr.amyprice@gmail.com; 2Department of Continuing Education, University of Oxford, Oxford, UK; 3Northern Ireland Methodology Hub, Belfast, UK; 4Northern Ireland Clinical Trials Unit, Belfast, UK; 5Centre for Public Health, Queen’s University Belfast, Belfast, UK
Conflict of Interest Disclosures:
Amy Price is a research fellow at and Sara Schroter is a full time employee of The BMJ. Mike Clarke reports involvement in many clinical trials and systematic reviews and has prepared and used supplementary material widely. He seeks funding for these trials and reviews, as well as for research into methodology, including dissemination and accessibility. No other disclosures were reported.
Acknowledgments:
We would like to acknowledge the 45 volunteers who piloted this research and shared valuable feedback to make the questions clear. The volunteers were community members, physicians, researchers, patients, and teachers. We thank all the researchers who completed the survey and especially those who shared comments. Their perspectives have increased our understanding.
