Conflict of Interest in Published Systematic Reviews on Interventions for 6 Common Clinical Diagnoses, 2010-2019
Abstract
Marek Czajkowski,1,2 Alexandra Snellman,1,2 Louise Olsson1,2
Objective
Conflicts of interest in systematic reviews (SRs) have gained recognition,1,2 but overall knowledge is limited, in particular from a clinical perspective. This study addresses financial conflict of interest (fCOI) statements in SRs relevant to primary care.
Design
A cross-sectional study of SRs published in 2010 and 2019 that evaluated interventions for 6 common diagnoses was designed. The diagnoses covered a substantial range of primary care specialties: chronic obstructive pulmonary disease, type 2 diabetes, hypertension, dementia, major depressive disorder, and osteoarthritis. PubMed was searched by a librarian in June 2021. Two independent researchers selected relevant SRs; any disagreements were resolved in consensus. Basic characteristics and data on fCOI and funding were extracted from full-text versions of SRs. The SRs of authors disclosing fCOI or reporting funding from the industry were categorized as “yes.” The SRs of authors declaring no fCOI and reporting no funding from the industry were categorized as “no.” Missing information either on authors´ disclosure of conflicting interests or information on funding of the SRs was categorized as “information missing.” A random sample of 50 SRs per fCOI category (“yes,” “no,” or “information missing”) was drawn, and data on fCOI and funding of the primary studies reported in these 150 SRs were extracted.
Results
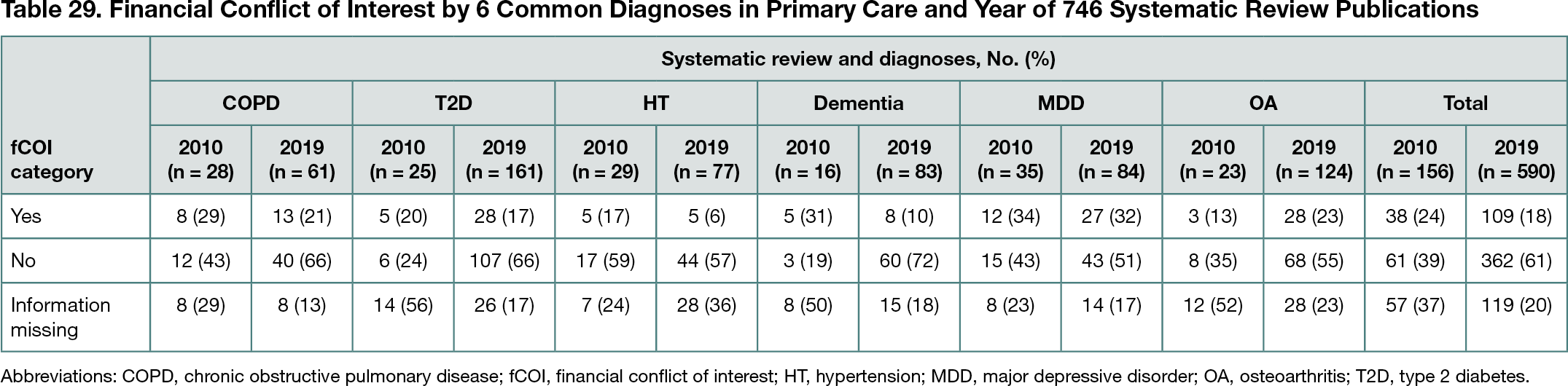
Of 2234 initially retrieved records, 746 (33%) relevant SRs were included. Diabetes and pharmacological interventions were the most common topics: 186 of 746 SRs (25%) for diabetes and 247 of 746 (33%) SRs for pharmacological interventions. Most often the first author was affiliated with an institution in China (130 [17%]). The number of relevant SRs increased 4-fold, from 156 in 2010 to 590 in 2019. In 2010, 38 (24%) SRs fulfilled the criteria for the fCOI “yes” category vs 109 (18%) in 2019 (Table 29). For osteoarthritis, the pattern was the opposite, with 3 (13%) categorized as “yes” in 2010 and 28 (23%) in 2019. The largest proportion of the fCOI “yes” category was noticed for depression, with 12 (34%) SRs in 2010 and 27 (32%) in 2019. In 2010, 57 (37%) SRs were categorized as “information missing” vs 120 (20%) in 2019. The only exception was for hypertension, with 7 (24%) SRs in 2010 vs 28 (36%) in 2019. Of the random subsample of 150 SRs, 30 (20%) reported data on fCOI or funding for the included primary studies.
Conclusions
The proportion of SRs disclosing fCOI or funding from industry was lower in 2019 compared with 2010 but was still missing in every fifth SR, with a direct clinical association with primary care. Risk factors for these shortcomings need to be identified, and given this, policies of journals are notable. Only a few SRs reported data on fCOI among the included primary studies.
References
1. Hakoum MB, Anouti S, Al-Gibbawi M, et al. Reporting of financial and non-financial conflicts of interest by authors of systematic reviews: a methodological survey. BMJ Open. 2016;6:e011997. doi:10.1136/bmjopen-2016-011997
2. Hansen C, Lundh A, Rasmussen K, Hróbjartsson A. Financial conflicts of interest in systematic reviews: associations with results, conclusions, and methodological quality. Cochrane Database Syst Rev. 2019;8(8):MR000047. doi:10.1002/14651858.MR000047.pub2
1School of Medical Sciences, Örebro University, Örebro, Sweden, marek.czajkowski@oru.se; 2Centre for Assessment of Medical Technology in Örebro, Örebro University Hospital, Örebro, Sweden
Conflict of Interest Disclosures
None reported.
Funding/Support
The project was supported financially by the Center for Clinical Research Dalarna, Falun, Sweden.
Role of the Funder/Sponsor
The funder did not have any role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the abstract; and decision to submit the abstract for presentation.
