Comparison of the Characteristics of COVID-19 and Non–COVID-19 Retractions
Abstract
Xiaoting Shi,1 Alison Abritis,2 Rujvee P. Patel,3 Mikas Grewal,4 Ivan Oransky,2,5,6 Joseph S. Ross,7,8,9 Joshua D. Wallach1
Objective
Concerns have been raised about the number of retracted COVID-19–related studies.1 Although this may be due to the greater scrutiny of COVID-19–related literature, little is known about the potential differences between retracted COVID-19–related and non–COVID-19–related studies, including author characteristics and reasons for retractions.
Design
In this cross-sectional analysis, all retractions of publications and withdrawals of preprints reporting the results of COVID-19–related and non–COVID-19–related medical studies indexed in the Retraction Watch Database between January 1, 2020, and May 5, 2022, were identified. Nonoriginal research (eg, retracted letters, viewpoints, and book chapters) and manuscripts classified by the Retraction Watch team as having been generated by companies that sell fake manuscripts (ie, paper mills) were excluded. For each retraction, the publication type (article or preprint) and design, number of authors, first and last authors and their affiliations, and date of retraction were recorded. The most prominent reasons for retraction were identified and grouped across similar categories. Scopus and then Google Scholar were searched to identify first and last author profiles, which were verified using affiliations, and we recorded an H-index and year of first publication for each author. The Fisher exact test and Mann-Whitney test were used to compare proportions of COVID-19–related vs non–COVID-19–related retractions.
Results
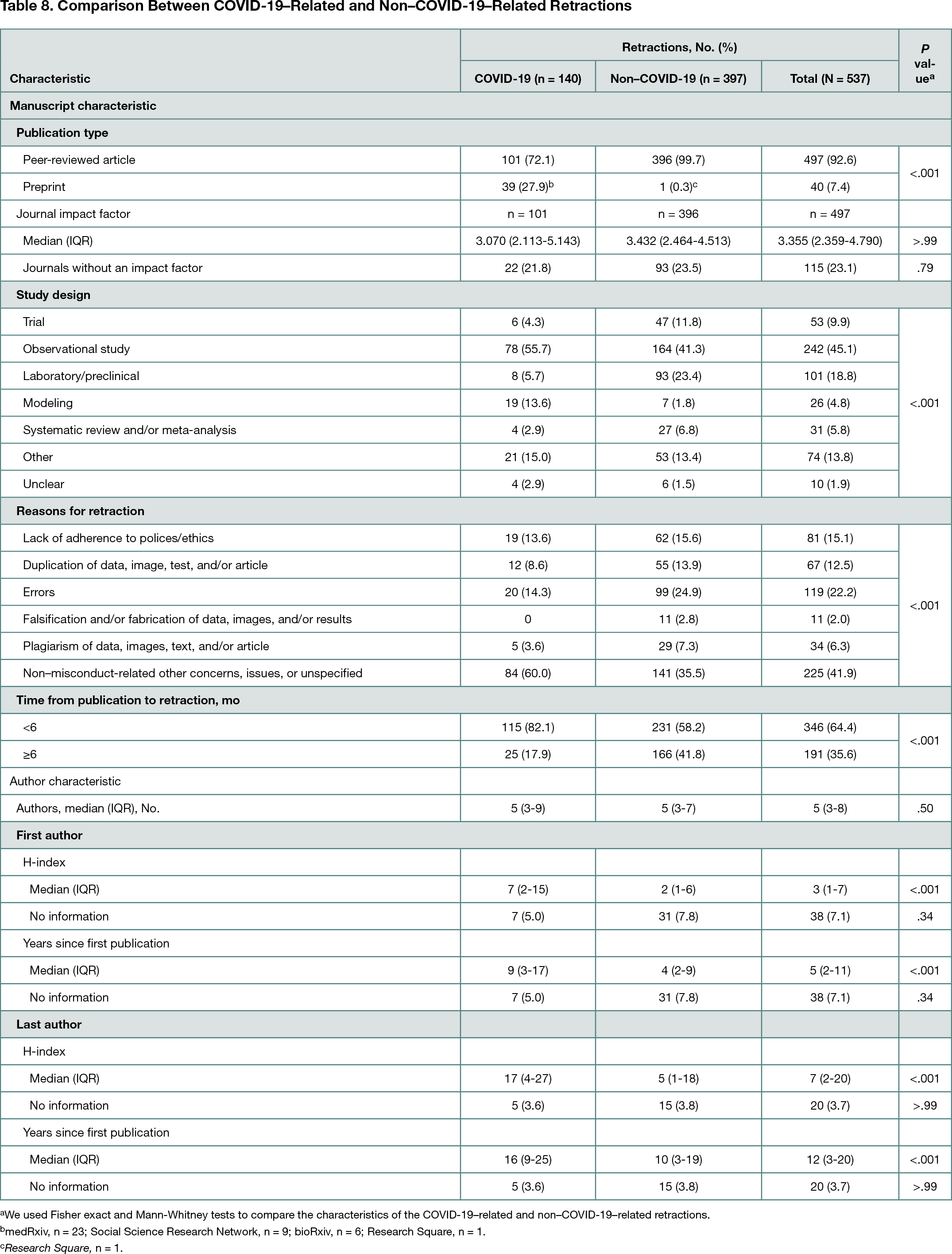
Between January 1, 2020, and May 5, 2022, 140 COVID-19–related and 397 non–COVID-19–related studies were indexed in the Retraction Watch Database (Table 8); 72.1% of the COVID-19–related studies were peer-reviewed articles (27.9% were preprints), whereas 99.7% of the non–COVID-19–related studies were peer-reviewed articles. COVID-19–related studies were more likely to be retracted or withdrawn within 6 months of publication or posting than non–COVID-19–related studies (82.1% vs 58.2%; P < .001). A greater proportion of modeling studies among the COVID-19–related than non–COVID-19–related studies was observed (13.6% vs 1.8%). COVID-19–related studies were more likely to be retracted without any explanation or to be removed for non–misconduct-related concerns than non–COVID-19–related studies (60.0% vs 35.5%). The first (29.1%) and last (30.7%) authors of COVID-19–related studies were more likely to have North American affiliations than the first (9.9%) and last (11.9%) authors of non–COVID-19–related studies. Nearly all first (97.6%) and last (98.0%) authors of all studies had academic or hospital affiliations. First and last authors of COVID-19–related studies had higher median (IQR) H-indexes than those of non–COVID-19–related manuscripts (7 [2-15] and 17 [4-27] vs 2 [1-6] and 5 [1-18]).
Conclusions
Author and manuscript characteristics differed between retracted COVID-19–related and non–COVID-19–related studies. Although there have been hundreds of COVID-19–related retractions since the start of the pandemic, there have also been tens of thousands of preprints and published articles.
Reference
1. Abritis A, Marcus A, Oransky I. An “alarming” and “exceptionally high” rate of COVID-19 retractions? Account Res. 2021;28(1):58-59. doi:10.1080/08989621.2020.1793675
1Department of Environmental Health Sciences, Yale School of Public Health, New Haven, CT, USA, joshua.wallach@yale.edu; 2Retraction Watch, The Center for Scientific Integrity, New York, NY, USA; 3Yale School of Public Health, New Haven, CT, USA; 4Yale School of Medicine, New Haven, CT, USA; 5Arthur Carter Journalism Institute, New York University, New York, NY, USA; 6Spectrum, The Simons Foundation, New York, NY, USA; 7Section of General Internal Medicine, Yale School of Medicine, New Haven, CT, USA; 8Yale-New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, CT, USA; 9Department of Health Policy and Management, Yale School of Public Health, New Haven, CT, USA
Conflict of Interest Disclosures
Xiaoting Shi is supported by the China Scholarship Council in the past 36 months. Alison Abritis is an employee of The Center For Scientific Integrity, which developed and maintains the Retraction Watch Database. Ivan Oransky is the volunteer executive director of The Center For Scientific Integrity, a nonprofit organization that is funded through database licensing fees, a subcontract from the University of Illinois on a Howard Hughes Medical Institute grant, and donations from individuals. Joseph S. Ross is a former associate editor of JAMA Internal Medicine, a current research editor at The BMJ, and receives research support through Yale University from Johnson and Johnson to develop methods of clinical trial data sharing, from the Medical Device Innovation Consortium as part of the National Evaluation System for Health Technology (NEST), from the US Food and Drug Administration (FDA) for the Yale-Mayo Clinic Center for Excellence in Regulatory Science and Innovation program (U01FD005938), from the Agency for Healthcare Research and Quality (R01HS022882), from the National Heart, Lung, and Blood Institute of the National Institutes of Health (NIH) (R01HS025164, R01HL144644), and from the Laura and John Arnold Foundation to establish the Good Pharma Scorecard at Bioethics International; in addition, he is an expert witness at the request of relator’s attorneys, the Greene Law Firm, in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against Biogen Inc. Joshua D. Wallach is supported by the FDA, Johnson & Johnson, and the National Institute on Alcohol Abuse and Alcoholism of the NIH under award 1K01AA028258.
