Registered Clinical Trial Trends in East Asia and the United States, 2014 to 2025
Abstract
Eun Hye Lee,1,2,3,4 San Lee,1,3,5 Jae Il Shin,3,4,6 John P. A. Ioannidis1,7,8,9
Objective
East Asia has become a major hub for clinical trials.1 Although a few studies have compared ClinicalTrials.gov with local registries in specific disease domains, comprehensive comparisons remain limited.2 This study analyzes registered clinical trial trends in East Asia during 2014 to 2025 and compares them with trends in the US.
Design
We extracted clinical trial data through the World Health Organization–operated International Clinical Trials Registry Platform (ICTRP), which integrates ClinicalTrials.gov and local registries. Because Taiwan’s registry is not part of the ICTRP, we collected its data separately. We analyzed overall clinical trial trends and filtered randomized clinical trials (RCTs), categorizing them by location, target size, and disease category. Trials conducted in only 1 country were categorized as domestic, whereas those conducted in 2 or more countries were classified as international. We explored the proportion of studies registered prospectively, defined as within 1 month of the trial start date.3
Results
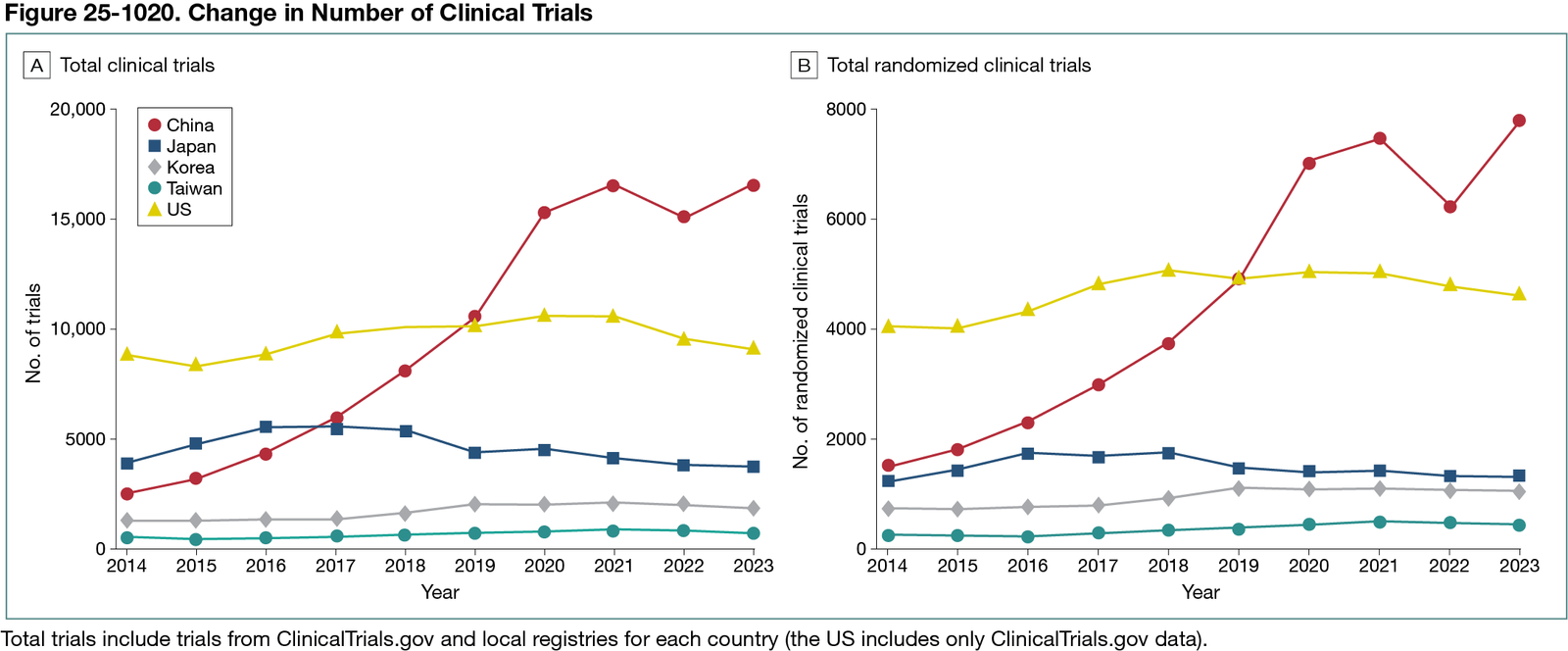
China experienced rapid growth in clinical trials, surpassing both Japan and the US in total trials and RCTs (Figure 25-1020). In China, the number of registered trials increased markedly from 2578 total trials (1521 RCTs) in 2014 to 16,612 (7798 RCTs) in 2023. In contrast, the US showed a more modest increase from 8841 trials (4056 RCTs) in 2014 to 9100 (4619 RCTs) in 2023. Except for China, all other countries experienced a postpandemic decline, with annual trial registrations decreasing by approximately 3% to 10% in recent years. Neoplastic diseases and cardiovascular and metabolic diseases accounted for the largest percentages, each representing approximately 15% to 25% of all registered trials. In the US, mental health trials increased from 11.9% in 2014 to 16.0% in 2023, reflecting growing interest in this area. In 2023, China’s RCTs were predominantly domestic, with international trials making up only 1.8% (140) of the total, while 13.1% (603) of RCTs in the US were international. Domestic RCTs were predominantly small-sized trials (<100 participants), while international RCTs were more commonly medium-sized (100-499 participants) or large-sized (≥500 participants) trials. In 2023, 75% of China’s RCTs (5841 of 7798) and 81% of Japan’s RCTs (1081 of 1339) were domestic trials registered in local registries, highlighting the predominant use of local registries in both countries. Although the trend has increased over the past decade, a considerable percentage of RCTs, ranging from 15% to 35% across countries, were still not registered prospectively in 2023. We recorded some inconsistencies and missing information across existing registries. Updated data to mid-2025 will be presented at the congress.
Conclusions
The study highlights the increasing prominence of East Asia, particularly China, in registered clinical trials, though with a primarily domestic focus. Improvements in existing trial registries covering East Asia are desirable.
References
1. Ali S, Egunsola O, Babar ZUD, Hasan SS. Clinical trials in Asia: a World Health Organization database study. Perspect Clin Res. 2019;10(3):121-124. doi:10.4103/picr.PICR_109_18
2. Doi M, Yukawa K, Sato H. Characteristics of Asian 4 countries on cancer clinical trials registered in the international clinical trials registry platform between 2005 and 2018. Chin Clin Oncol. 2021;10(3):28. doi:10.21037/cco-21-17
3. Klatte K, Sluka c, Gloy V, et al. Towards full clinical trial registration and results publication: longitudinal meta-research study in Northwestern and Central Switzerland. BMC Med Res Methodol. 2023;23(1):27. doi:10.1186/s12874-023-01840-9
1Meta-Research Innovation Center at Stanford (METRICS), Stanford University, Stanford, CA, US, jioannid@stanford.edu; 2Division of Pulmonology, Allergy and Critical Care Medicine, Department of Internal Medicine, Yongin Severance Hospital, Yonsei University College of Medicine, Yongin-si, Gyeonggi-do, South Korea; 3The Center for Medical Education Training and Professional Development, Yonsei Donggok Medical Education Institute, Seoul, Korea; 4Severance Underwood Meta-Research Center, Institute of Convergence Science, Yonsei University, Seoul, South Korea; 5Department of Psychiatry and the Institute of Behavioral Science in Medicine, Yonsei University College of Medicine, Seoul, South Korea; 6Department of Pediatrics, Yonsei University College of Medicine, Seoul, South Korea; 7Stanford Prevention Research Center, Department of Medicine, Stanford University School of Medicine, Stanford, CA, US; 8Department of Epidemiology and Population Health, Stanford University School of Medicine, Stanford, CA, US; 9Department of Biomedical Data Science, Stanford University School of Medicine, Stanford, CA, US.
Conflict of Interest Disclosures
John P. A. Ioannidis is a member of the Peer Review Congress Advisory Board but was not involved in the review or decision for this abstract.
Funding/Support
The work of John P. A. Ioannidis is supported by an unrestricted gift from Sue and Bob O’Donnell to Stanford University.
Role of the Funder/Sponsor
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information
A language model based on Bidirectional Encoder Representations from Transformers (BERT) was employed on December 30, 2024, to classify disease categories in RCTs. Specifically, PubMedBERT, a version pretrained on biomedical literature from PubMed, was used to automate the disease classification of clinical trials. San Lee takes responsibility for the integrity of the content. Eun Hye Lee and San Lee contributed equally to this study.
