Public Access to Information Cited in Rare Disease Reports
Abstract
Mengyuan Fu,1,2 Kexin Ling,3 Xinyi Zhou,3 Sneha Dave,4 Can Li,3 Luwen Shi,2,3 Xiaodong Guan,2,3 Joseph S. Ross1,5,6
Objective
This study aims to examine whether the information sources cited within National Organization for Rare Disorders (NORD) rare disease reports were publicly available, as well as any associated costs to purchase access, given that patients, caregivers, and clinicians may rely on these sources for critical information to inform their clinical decision-making.
Design
We randomly selected 20% of all NORD rare disease reports available as of February 2024.1 Citations within these reports were classified into 5 source types, including publications in scholarly journals (research articles, case reports, reviews), textbooks, and other internet resources. Each citation was searched using Google and categorized as open access (freely available online), accessible at cost, or unavailable online. For citations with associated costs to purchase access, we recorded the lowest listed price before taxes. A 10% cross-sample validation was conducted between each pair of researchers to check consistency. All data were collected between April and May, 2024. We used descriptive statistics to calculate the proportion of citations categorized by availability, overall and by year and source type, as well as median costs, overall and by source type. We used χ² tests to evaluate differences in open access rates by year and source type. A 2-sided P < .05 was considered statistically significant. Analyses were performed using Stata MP version 16.0.
Results
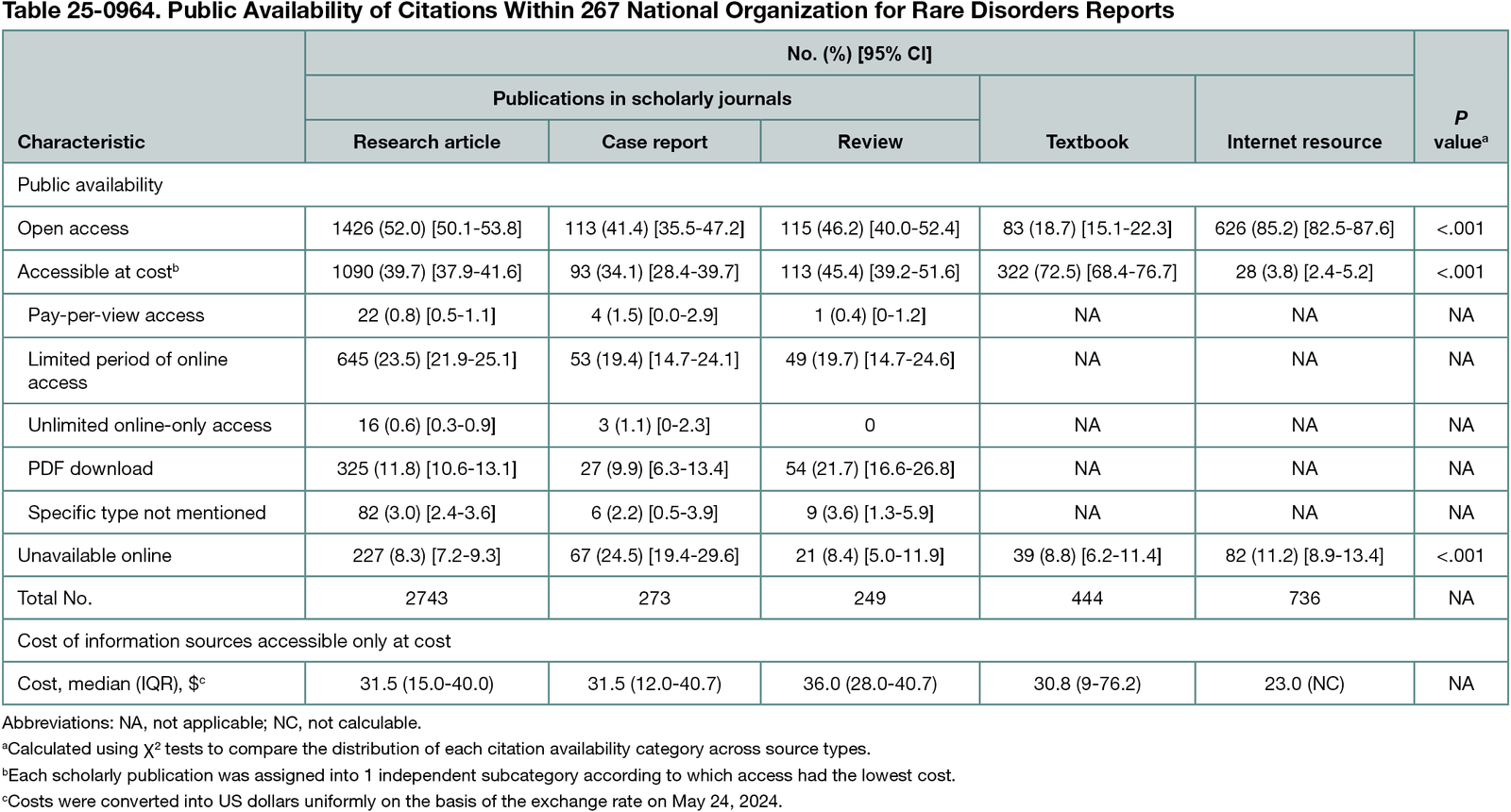
Among 1333 NORD rare disease reports available as of February 2024, 267 were randomly selected for study. These 267 reports included 4445 citations to 3506 unique information sources; the median (IQR) number of citations per report was 15 (11-20), including 9 (5-13) research articles, 0 (0-1) case reports, 0 (0-0) review articles, 1 (0-3) textbook, and 2 (1-4) other internet resources. Among 3506 unique information sources cited, 1750 (49.9%) were open access, 1381 (39.4%) were accessible at cost, and 375 (10.7%) were unavailable online (Table 25-0964). Among citations accessible only at cost, the median (IQR) cost was $31.5 ($15.0-$40.0), whereas the total cost per report for all citations accessible only at cost was $167.7 ($82.0-$258.7). The overall proportion of citations published in scholarly journals available as open access increased from 30.0% (258/860) prior to 2001 to 79.8% (95/119) from 2021 onwards (P < .001).
Conclusions
While NORD should be applauded for making its rare disease reports publicly available, we found that half of the information sources cited by these reports were not open access, potentially limiting equitable access to critical information for rare disease communities.
Reference
1. National Organization for Rare Disorders. Rare disease database. Accessed March 1, 2024. https://rarediseases.org/rare-diseases/
1Department of Internal Medicine, Yale School of Medicine, New Haven, CT, US, joseph.ross@yale.edu; 2International Research Center for Medicinal Administration, Peking University, Beijing, China; 3Department of Pharmacy Administration and Clinical Pharmacy, School of Pharmaceutical Sciences, Peking University, Beijing, China; 4Generation Patient, Indianapolis, IN, US; 5Center for Outcomes Research and Evaluation, Yale–New Haven Health System, New Haven, CT, US; 6Department of Health Policy and Management, Yale School of Public Health, New Haven, CT, US.
Conflict of Interest Disclosures
Mengyuan Fu currently receives research support from the National Natural Science Foundation of China (72304011). Sneha Dave is the founder and executive director of Generation Patient, an organization that receives grant funding from Arnold Ventures, Commonwealth Fund, Disability Inclusion Fund, Third Wave Fund, Responsible Technology Youth Power Fund, the Helmsley Charitable Trust, Patient-Centered Outcomes Research Institute, and Lucile Packard Foundation for Children’s Health. Joseph S. Ross currently receives research support through Yale University from Johnson and Johnson to develop methods of clinical trial data sharing, from the Food and Drug Administration for the Yale-Mayo Clinic Center for Excellence in Regulatory Science and Innovation (CERSI) program (U01FD005938), from the Agency for Healthcare Research and Quality (R01HS022882), and from Arnold Ventures; formerly received research support from the Medical Device Innovation Consortium as part of the National Evaluation System for Health Technology (NEST) and from the National Heart, Lung and Blood Institute of the National Institutes of Health (NIH) (R01HS025164, R01HL144644); was an expert witness at the request of Relator’s attorneys, the Greene Law Firm, in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against Biogen Inc. that was settled in September 2022; and is a deputy editor for JAMA. No other disclosures were reported.
