Noninferiority vs Superiority Trials in Cardiovascular Research: Trends, Success in Meeting the Primary Outcome, and Engagement Patterns
Abstract
Ashkan Hashemi,1 Isaac Dreyfus,2 Nicholas Varunok,3 John Burton,4 Sina Rashedi,5 Farbod Zahedi Tajrishi,6 Behnood Bikdeli5
Objective
The role of noninferiority trials in cardiovascular research remains unclear. We sought to determine the proportion of noninferiority vs superiority trials, their likelihood of meeting primary outcomes, and their academic impact and online engagement.
Design
We reviewed cardiovascular randomized controlled trials (RCTs) published in The New England Journal of Medicine, JAMA, and The Lancet between January 2014 and December 2019. Cardiovascular trials were defined based on review by 2 independent reviewers for topics that included patients with cardiovascular diseases or tested cardiovascular outcomes. Trials were categorized by design (superiority vs noninferiority), and assessed for primary outcome success, academic impact (Google Scholar citations), and online engagement (Altmetric Attention Score). Linear trends were assessed via linear regression. An interaction term was tested between trial design (superiority vs noninferiority) and outcome success (meeting the primary outcome or not) with respect to citation count.
Results
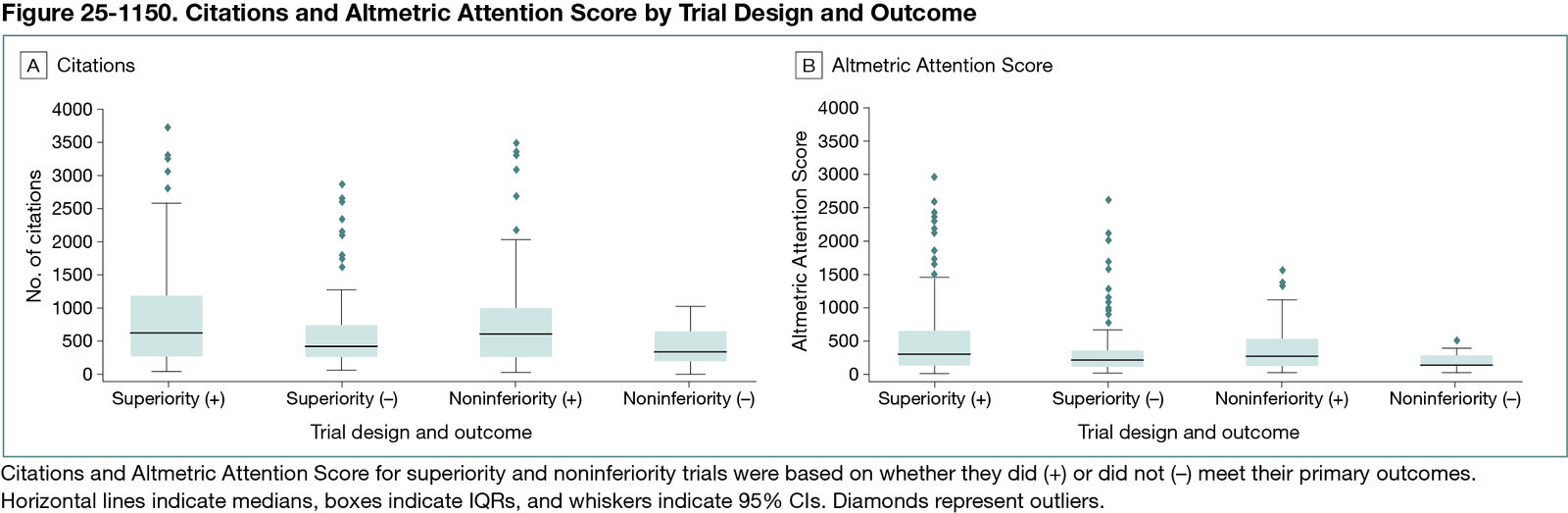
Among 429 cardiovascular RCTs, 100 (23.3%) were noninferiority trials, with their proportion increasing over time (P = .009). Noninferiority trials were significantly more likely to meet their primary outcome than superiority trials (79% vs 54%; P < .001). Median citations were comparable between noninferiority and superiority trials (560 [IQR, 248-851] and 505 [IQR, [274-886], respectively; P = .83), as were Altmetric Attention Scores (226 [IQR, 124-481] and 234 [IQR, 120-542], respectively; P = .70) (Figure 25-1150). Regardless of design, trials that met their primary outcome had higher citation counts than those that did not (superiority trials, 615 [IQR, 280-1200] vs 423 [IQR, 267-734], respectively; P = .003; noninferiority trials, 613 [IQR, 261-998] vs 338 [IQR, 201-635], respectively; P = .049). There was no significant interaction between trial design and outcome success with respect to citation counts (P for interaction = .87).
Conclusions
Noninferiority trials are increasingly shaping cardiovascular research and are more likely to meet their primary outcome, with academic engagement and online attention comparable to superiority trials. The higher success rate observed in noninferiority trials may, in part, reflect the discretionary—and at times overly permissive—approach to noninferiority margin selection, warranting further study. Finally, Altmetric Attention Scores reflect online attention but not clinical impact; future work will assess influence on guidelines and US Food and Drug Administration labeling.
1Weill Cornell Medicine, New York, NY, US; 2David Geffen School of Medicine, University of California, Los Angeles, CA, US; 3Vanderbilt University Medical Center, Nashville, TN, US; 4Keck School of Medicine, University of Southern California, Los Angeles, CA, US; 5Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, US, bbikdeli@bwh.harvard.edu; 6Tulane University School of Medicine, New Orleans, LA, US.
Conflict of Interest Disclosures
None reported.
