Development of a Tool for Addressing Conflicts of Interest in Trials (TACIT) for Use in Systematic Reviews
Abstract
Andreas Lundh,1,2,3 Isabelle Boutron,4,5,6 Lesley A. Stewart,7 Asbjørn Hróbjartsson1,2
Objective
Industry funding and authors’ financial conflicts of interest are common in clinical trials and associated with favorable trial conclusions and seemingly greater treatment effects. However, there has been little guidance on how to take funding and conflicts of interest into account in evidence synthesis. To address this, we developed the Tool for Addressing Conflicts of Interest in Trials (TACIT).
Design
We established an international working group of 20 experts in trial and systematic review methods, conflicts of interest, and biomedical publishing. Four substudies informed tool development. First, a systematic review found that current study appraisal tools address funding and conflicts of interest superficially.1 Second, an interview study of experienced trial researchers found that funding and conflicts of interest may unduly influence trials through various mechanisms but influence may be minimized if properly managed.2 Third, a cross-sectional study of trials included in Cochrane reviews found that searching beyond trial publications using a structured information retrieval approach may improve identification of trial funding and researchers’ conflicts of interest.3 Fourth, an interview and questionnaire study of expected users piloting a preliminary version of TACIT found that users generally had a positive experience but suggested more guidance on ease of use and how to deal with insufficient information on conflicts of interest. The tool has been iteratively revised mainly based on feedback from the working group and expected users. A final consensus meeting was held in May 2025, where the result of substudies, themes for discussion, and a near-final version of the tool were presented. Based on the consensus meeting, the tool will be revised and made public together with a Microsoft Excel–based application after summer 2025.
Results
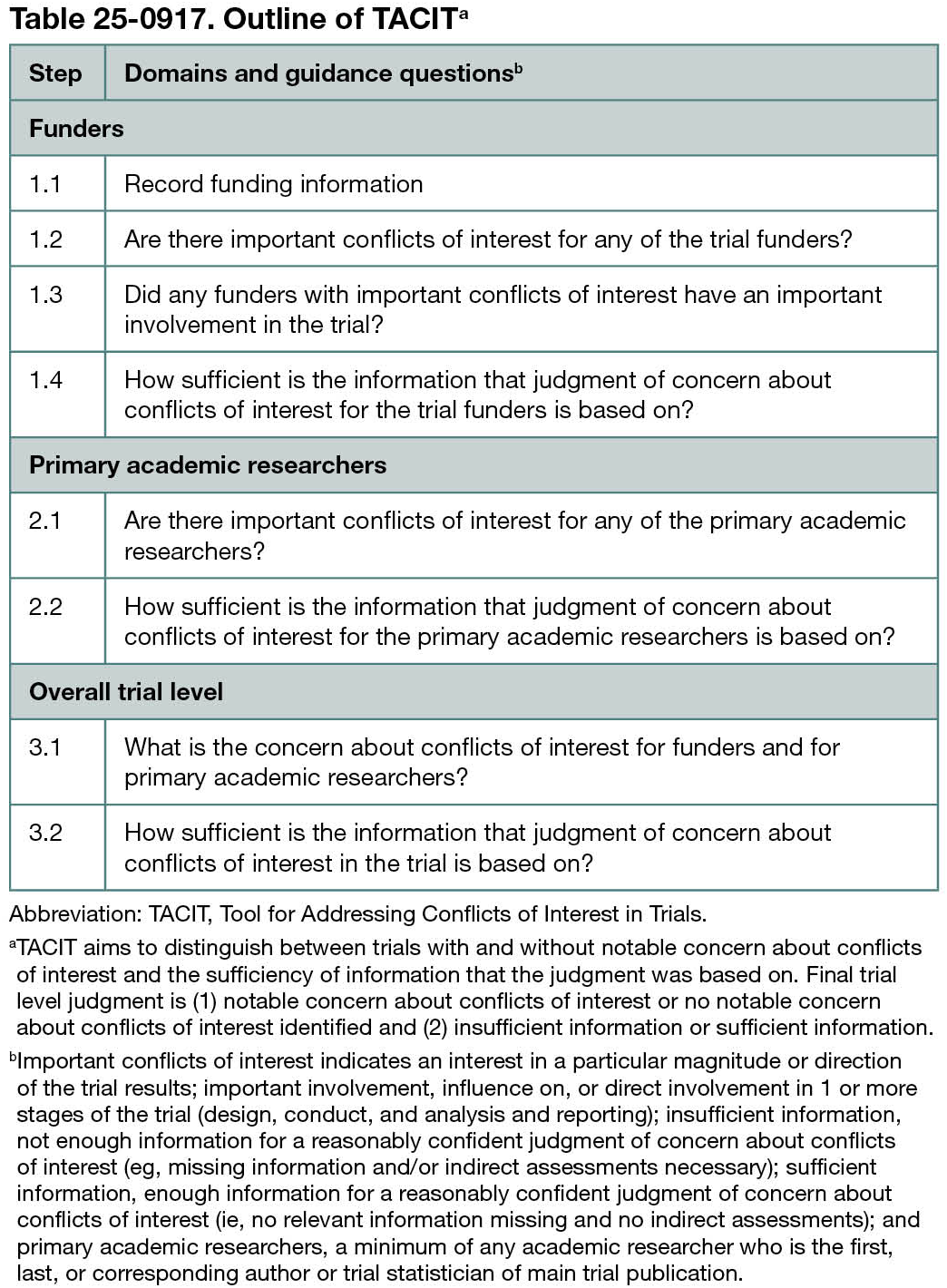
Through a series of guidance questions, the tool facilitates a judgment of notable concern about conflicts of interest and the sufficiency of information that the judgment of concern was based on (Table 25-0917). The judgment involves 3 levels: trial funders, the trial’s primary academic researchers, and overall trial level. The tool provides an overview of funding and conflicts of interest information for trials included in the systematic review, and it categorizes trials into trials with or without notable concern about conflicts of interest. Results of the assessments may be incorporated into review syntheses using subgroup or sensitivity analysis.
Conclusions
TACIT provides a structured, coherent, and transparent assessment of conflicts of interest in trials included in systematic reviews. The tool may be used by systematic reviewers, guideline panels, and other users undertaking evidence syntheses.
References
1. Lundh A, Rasmussen K, Østengaard L, Boutron I, Stewart LA, Hróbjartsson A. Systematic review finds that appraisal tools for medical research studies address conflicts of interest superficially. J Clin Epidemiol. 2020;120:104-115. doi:10.1016/j.jclinepi.2019.12.005
2. Østengaard L, Lundh A, Tjørnhøj-Thomsen T, et al. Influence and management of conflicts of interest in randomised clinical trials: qualitative interview study. BMJ. 2020;371:m3764. doi:10.1136/bmj.m3764
3. Faltinsen E, Todorovac A, Boutron I, Stewart LA, Hróbjartsson A, Lundh A. A structured approach to information retrieval improved identification of funding and researchers’ conflicts of interest in trials included in Cochrane reviews. J Clin Epidemiol. 2023;161:104-115. doi:10.1016/j.jclinepi.2023.06.020
1Cochrane Denmark & Centre for Evidence-Based Medicine Odense (CEBMO), Department of Clinical Research, University of Southern Denmark, Odense, Denmark, alundh@health.sdu.dk; 2Open Patient data Exploratory Network (OPEN), Odense University Hospital, Odense, Denmark; 3Department of Respiratory Medicine and Infectious Diseases, Copenhagen University Hospital—Bispebjerg and Frederiksberg, Copenhagen, Denmark; 4Université Paris Cité and Université Sorbonne Paris Nord, Inserm, INRAe, Centre for Research in Epidemiology and Statistics (CRESS), Paris, France; 5Centre d’Épidémiologie Clinique, Hôpital Hôtel Dieu, Université Paris Cité, Paris, France; 6Cochrane France, Paris, France; 7Centre for Reviews and Dissemination, University of York, York, UK.
Conflict of Interest Disclosures:
Isabelle Boutron is a member of the Peer Review Congress Advisory Board but was not involved in the review or decision for this abstract. No other disclosures were reported.
Additional Information:
The authors are members of the Tool for Addressing Conflicts of Interest in Trials (TACIT) Steering Group. We thank members of the TACIT working group and coauthors of TACIT substudies for their contribution to the development of TACIT.
