COVID-19 Public Health Scientific Publications From the Centers for Disease Control and Prevention, January 2020 to January 2022
Abstract
Elissa Meites,1 Martha Knuth,1 Kaely Hall,1 Elizabeth Stephenson,1 Patrick Dawson,1 Teresa W. Wang,1 Wei Yu,1 Muin Khoury,1 Barbara Ellis,1 Brian A. King1
Objective
High-quality scientific evidence is critical to support public health decision-making. During public health emergencies, including the COVID-19 pandemic, the US Centers for Disease Control and Prevention (CDC) has been an important source of scientific information. The CDC can provide timely data on and evidence from fundamental public health activities, including domestic and international epidemiologic investigations, laboratory detection, disease surveillance and research, and other scientific work.1 Routinely, all manuscripts authored by CDC scientists are reviewed internally for scientific quality before publication through a rigorous multilevel process involving subject matter experts and other CDC staff.2 The objective of this analysis was to understand the CDC’s contributions to new COVID-19 science.
Design
A bibliometric analysis was conducted of the CDC’s COVID-19 public health scientific publications and their effect from January 20, 2020, to January 20, 2022, using a quality improvement approach (SQUIRE, version 2.0). COVID-19 scientific articles were cataloged if they had at least 1 CDC-affiliated author, had been reviewed and cleared internally, and were subsequently published in a peer-reviewed scientific journal and indexed in the World Health Organization’s COVID-19 database. The priority topics addressed were identified according to the CDC’s COVID-19 Public Health Science Agenda and were assigned tags using keyword scripts in EndNote, and their scientific effects were assessed using Scopus citations, news media, social media impressions, and Altmetric attention scores. This work received a nonresearch determination from the CDC.
Results
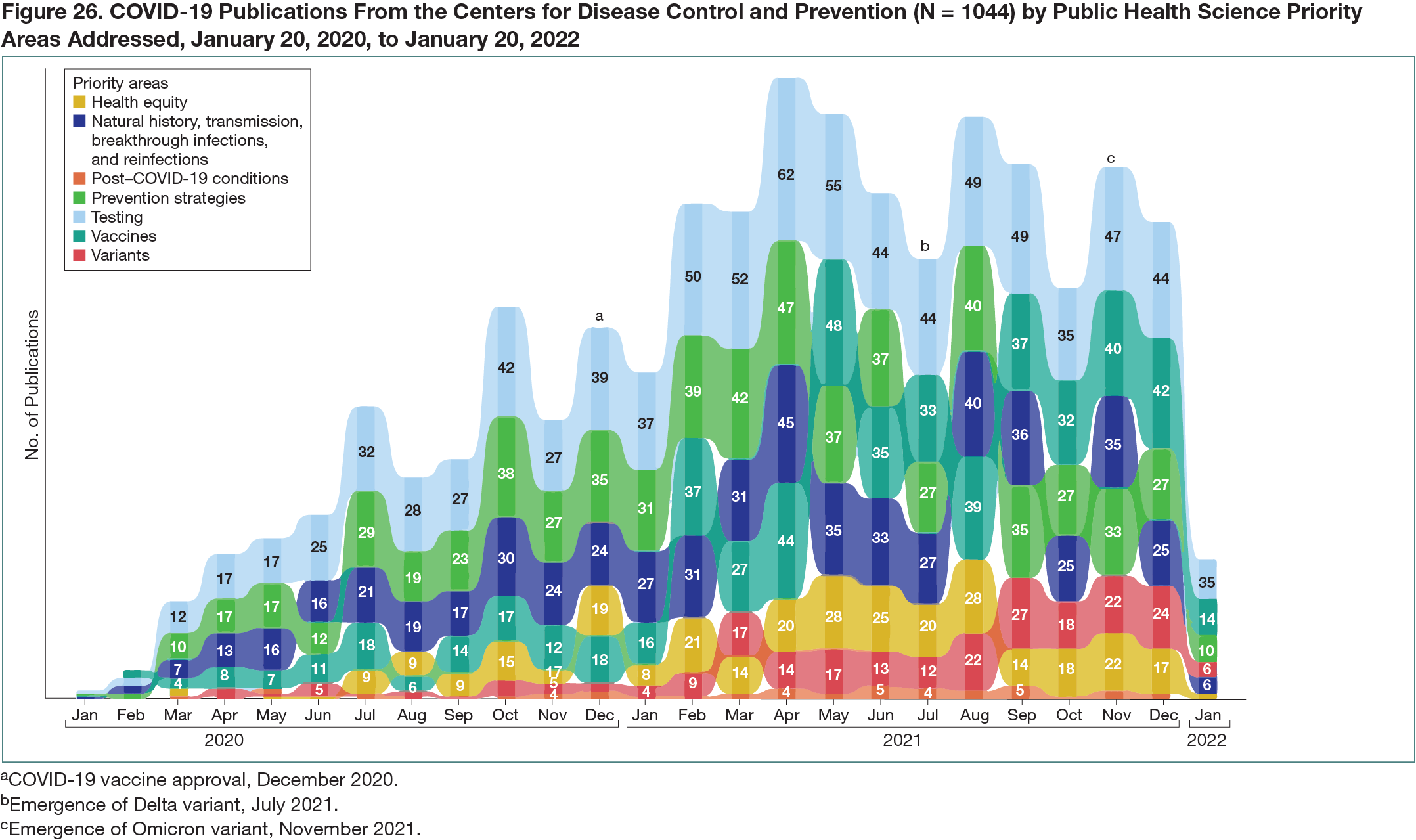
During the first 2 years of the pandemic, CDC authors contributed to 1044 unique COVID-19 scientific publications in 208 journals. The topic areas of focus commonly included testing (853 publications [82%]); prevention strategies (658 publications [63%]); natural history, transmission, breakthrough infections, and reinfections (587 publications [56%]); vaccines (567 publications [54%]); health equity (308 publications [30%]); variants (232 publications [22%]); and post–COVID-19 conditions (44 publications [4%]) (Figure 26). In addition, the CDC adapted to changing key scientific questions as the pandemic evolved, addressing issues including COVID-19 testing expansions; COVID-19 vaccine effectiveness, safety, duration of protection, and access; emergence of new viral variants of concern; and improving health equity.3 These publications were cited 40,427 times in the scientific literature, have been the topic of 81,921 news media reports and 1,058,893 social media impressions, and received a combined total Altmetric attention score of 920,763. Preprints were not included in this analysis.
Conclusions
Throughout the COVID-19 pandemic, the CDC has supported the development of high-quality science oriented to improving public health outcomes. The lessons learned from this evaluation included: (1) the agency’s COVID-19 Public Health Science Agenda helped guide scientific activities; (2) scientific manuscripts developed, reviewed, and published to address priority topics became highly effective; and (3) the CDC is committed to monitoring emerging issues and addressing gaps in evidence needed to improve health outcomes. Data-driven strategies are essential to address disparities and improve the health outcomes of people disproportionately affected by COVID-19.
References
1. Bunnell R, Ryan J, Kent C; CDC Office of Science, CDC Excellence in Science Committee. Toward a new strategic public health science for policy, practice, impact, and health equity. Am J Public Health. 2021;111(8):1489-1496. doi:10.2105/AJPH.2021.306355
2. Cono J, Jaffe H. The CDC clearance process: supporting quality science. Am J Public Health. 2015;105(6):e1-e2. doi:10.2105/AJPH.2015.302691
3. Cohn AC,. One year of COVID-19 vaccines: a shot of hope, a dose of reality. JAMA. 2022;327(2):119-120. doi:10.1001/jama.2021.23962
1Chief Science Office, COVID-19 Response, Centers for Disease Control and Prevention, Atlanta, GA, USA, emeites@cdc.gov
Conflict of Interest Disclosures
None reported.
Funder/Sponsor
The Centers for Disease Control and Prevention (CDC).
Role of the Funder/Sponsor
The CDC participated in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the abstract; and decision to submit the abstract for presentation.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the CDC.
Additional Information
Martha Knuth is a co-corresponding author.
