Characterizing Adverse Event Methods Reported in ClinicalTrials.gov and Publications
Abstract
Kyungwan Hong,1 Mark Basista,1 Tony Tse1
Objective
Information about how adverse events (AEs) were specified, collected, and assessed for clinical trials (ie, AE methods) is essential to accurately interpret the safety profiles of studied interventions.1 ClinicalTrials.gov provides a structured format for trialists to report aggregate results information and AE methods that complement the published literature. Prior work compared reporting of AEs in publications and ClinicalTrials.gov in terms of completeness and consistency,2 examined differences between sources in reporting AE methods for systematic collection,3 and assessed AE methods reporting in publications and identified methodologic weaknesses.1 This cross-sectional analysis explored the extent that AE methods reported in ClinicalTrials.gov complemented corresponding publications.
Design
ClinicalTrials.gov was searched on August 1, 2024, for drug and biologic trials with primary completion dates between January 18, 2017, and August 1, 2021, to allow 3 years for results posting and publication. A total of 9659 records were initially retrieved. After randomization, the records were reviewed sequentially until a convenience sample of 40 trials with summary results posted on ClinicalTrials.gov and corresponding primary results articles in PubMed were identified. The identified trials were then analyzed. All authors reviewed and extracted text for 5 ClinicalTrials.gov AE method data elements: collection period (time frame), vocabulary source for AE terms (how AE terms were standardized), collection approach (systematic or nonsystematic), analysis population, and whether participants withdrew due to harms. Each set of trial results was independently reviewed by 2 authors, with discrepancies resolved by consensus.
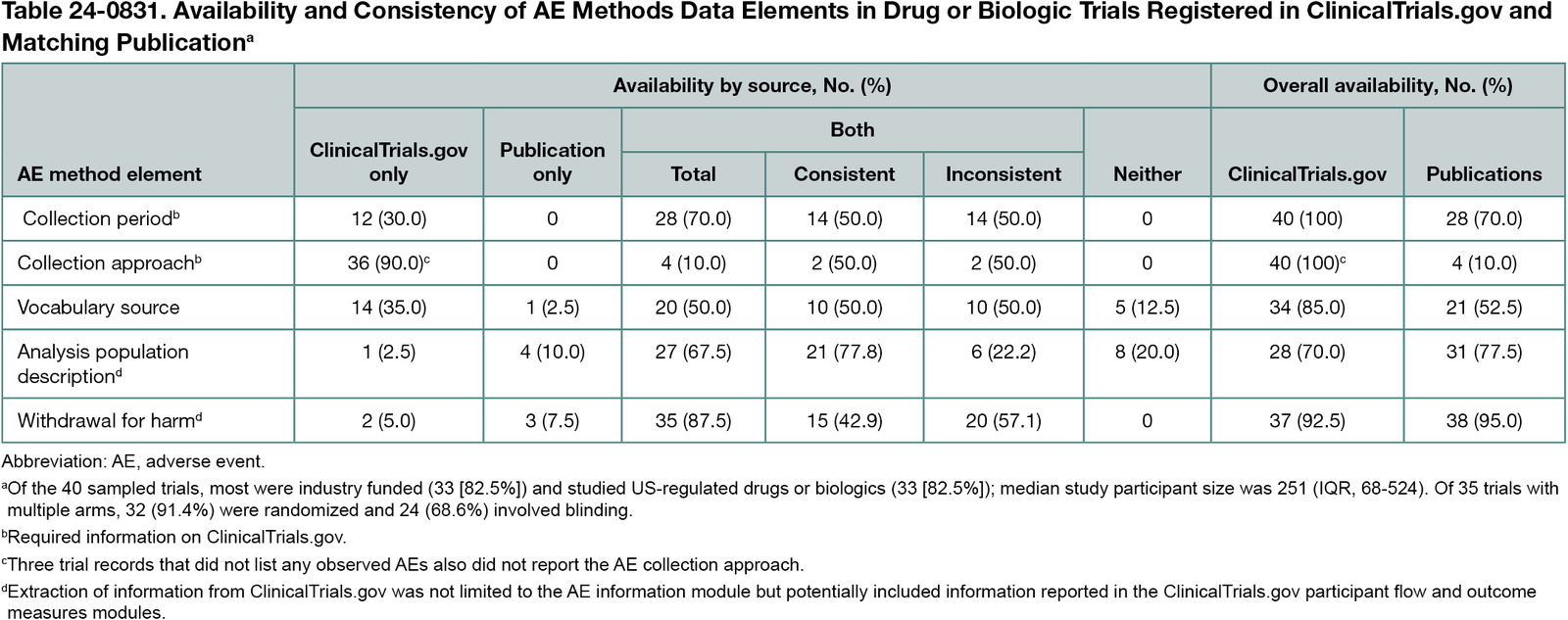
Results
Collection period (28 [70.0%]), analysis population (27 [67.5%]), and withdrawal for harm (35 [87.5%]) were described for over half of the 40 sampled trials in ClinicalTrials.gov and/or publications (Table 24-0831). However, consistency varied; for example, of the 35 trials with number of participants withdrawn for harm described in both sources, 20 (57.1%) provided inconsistent information. Collection approach was described in both sources infrequently (4 [10.0%]). Other AE methods (eg, vocabulary source) were reported for some trials in only one or neither source.
Conclusions
A few AE methods were not reported on ClinicalTrials.gov and/or publications, indicating a need for improvement. Complete AE methods reporting is needed for accurate safety profiles of study interventions and unbiased risk-benefit assessments. Even when available, reported AE methods were frequently inconsistent between sources, potentially due to variations in time frames used for AE reporting. Limitations included excluding supplementary appendices and study protocols that may provide additional methodologic details on AE methods. Future research is needed to review publicly available study protocols for AE methods information, compare the quality of AE methods reporting by source, and characterize trials in which AE methods have and have not been completely reported (eg, by funding source, study design).
References
1. Phillips R, Hazell L, Sauzet O, Cornelius V. Analysis and reporting of adverse events in randomised controlled trials: a review. BMJ Open. 2019;9(2):e024537. doi:10.1136/bmjopen-2018-024537
2. Krešo A, Grahovac M, Znaor L, Marušić A. Safety reporting in trials on glaucoma interventions registered in ClinicalTrials.gov and corresponding publications. Sci Rep. 2024;14(1):27762. doi:10.1038/s41598-024-79394-z
3. Mayo-Wilson E, Fusco N, Li T, Hong H, Canner JK, Dickersin K; MUDS Investigators. Harms are assessed inconsistently and reported inadequately part 1: systematic adverse events. J Clin Epidemiol. 2019;113:20-27. doi:10.1016/j.jclinepi.2019.04.022
1National Center for Biotechnology Information, National Library of Medicine, National Institutes of Health, Bethesda, MD, US, kyungwan.hong@nih.gov.
Conflict of interest Disclosures
Kyungwan Hong reported being employed by the National Institutes of Health (NIH) as a policy analyst with the ClinicalTrials.gov program. Mark Basista reported conducting this work while contracting with ICF. Tony Tse reported being employed by the NIH as an analyst with the ClinicalTrials.gov program. No other disclosures were reported.
Funding/Support
This work was supported by the National Center for Biotechnology Information of the National Library of Medicine, NIH.
Role of the Funder/Sponsor
The funder was not directly involved in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the abstract for presentation.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily reflect the views or policies of the NIH.
